Overview | Principle | Indication | Sampling | Performance | Your benefit | Literature | Availability

In a nutshell
- Epigenetic DNA methylation test for the detection of all carcinomas originating from the uterus, especially endometrial carcinoma, but also endocervical carcinoma.
- The test is indicated in peri- and postmenopausal women with abnormal uterine bleeding.
- Sample collection by cervicovaginal swab.
- 95% less false positives compared to transvaginal sonography.
- The test reduces the number of women who have to undergo hysteroscopy with curettage by 90%.
- A negative WID®-easy test result indicates a high probability that there is no malignancy, so that conservative treatment and active monitoring are an alternative to hospital referral for many patients.
The principle of the WID®-easy test
Indication
Test for simple and rapid screening of women presenting with symptoms suggestive of endometrial cancer. These are particularly peri- and postmenopausal women with abnormal uterine bleeding.
The procedure
A cervicovaginal swab is sent to the diagnostic laboratory.
After DNA extraction and bisulfite conversion, a quantitative PCR reaction is performed.
The WID®-easy test, which is referred to in the scientific literature as “WID-qEC test”, measures the methylation status of DNA regions of the GYPC and ZSCAN12 genes, which are known to be highly methylated in endometrial cancer and cervical cancer.
These measured values (indicated in “PMR” = “Percentage of fully Methylated Reference”) are summed to give “∑ PMR”. The result of the test depends on the determined value ∑ PMR and is provided to the physician in the test report.
Sources: www.eutops.institute and Evans et al, The Lancet Oncology, 2023
The test result
The test result will be available to you in a few days. Please ask your laboratory for the current processing times (sample receipt in the laboratory until the report is issued) for the WID®-easy test.
The report states the ∑PMR value determined and an interpretation of the result based on Evans et al., 2023. The formulation of the test result is the responsibility of the healthcare facility that produces and uses the test.
The sampling*
Sampling is carried out using a Copan swab and medium.

Copan Swab and Medium
Sampling is carried out using the Copan FLOQswab™ (552C.80PB) and the eNAT medium (608C) from Copan, which also offers a corresponding kit (608CS01R). Both will be provided to you by your laboratory.
The procedure
Do not use any lubricant before taking the sample for the WID®-easy test.
The sample collection for the WID®-easy test takes place
- before taking a sample for another test, e.g., for a PAP smear.
- before inserting another substance into the vagina or near the cervix, such as acetic acid, for visual inspection (VIA).
- before transvaginal sonography
- or at the earliest three days after these measures.
Detailed instructions on sampling can be found at the following link*.
Limitations
The WID®-easy test detects the methylation patterns of tumor DNA indicative of endometrial cancer.
Conditions that restrict the drainage of tumor DNA from the uterine cavity into the vagina, such as large endocervical polyps or fibroids, may affect the sensitivity of the test.
The patient should not have vaginal intercourse without using a condom for at least 24 hours before taking the WID®-easy test.
* The specific instructions for sampling are the responsibility of the healthcare facility that produces and uses the test. These instructions will be provided by the health institution. The example presented here is a current working hypothesis.
S3 guideline endometrial cancer

Diagnostic pathway for patients with postmenopausal bleeding
According to the current S3 guideline on endometrial carcinoma (AWMF register number: 032/034-OL, September 2022), transvaginal sonography as well as a cytological examination, are recommended after clinical examination.
Transvaginal sonography determines the endometrial thickness and, if the thickness is ≥ 3 mm, surgical, diagnostic procedures such as Tao Brush™, Pipelle™, hysteroscopy and curettage are recommended.
In women with peri- and postmenopausal abnormal bleeding, this threshold is reached or exceeded in over 50% of cases.
(Evans et al., The Lancet Oncology, 2023).
The performance of the WID®-easy test compared to the transvaginal sonographic examination
The performance of the WID®-easy test compared to the transvaginal ultrasound examination
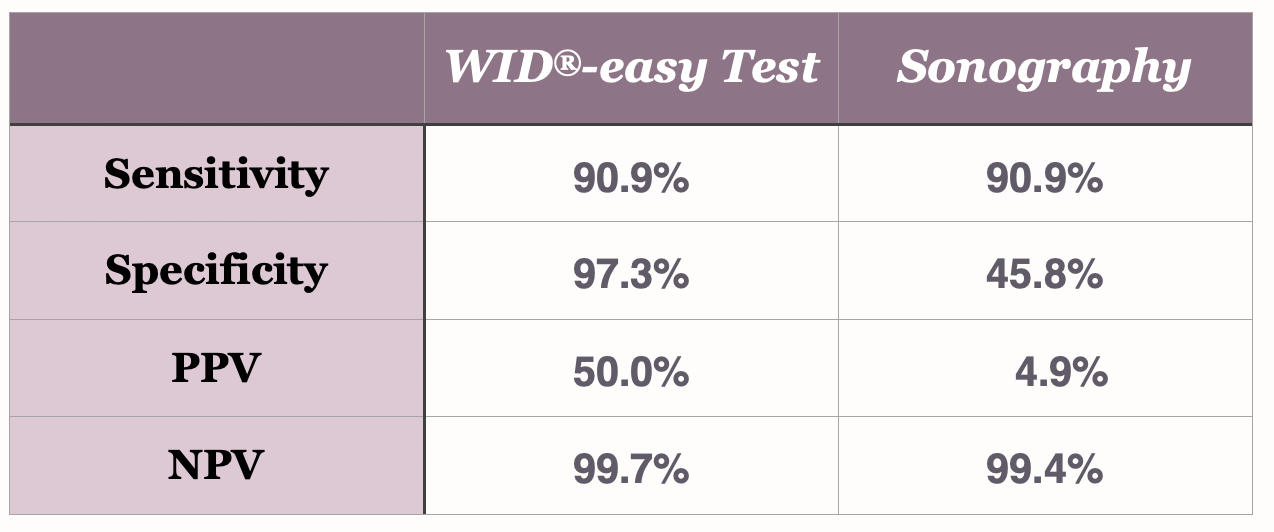
The WID®-easy test is characterized by extremely high values for sensitivity (90.9%), specificity (97.3%) and NPV (99.7%).
Using the ∑PMR cut-off value of 0.3, the PPV of the WID®-easy test is 50 %, ten times higher than that of sonography, whose PPV is only 5 %.
(Evans et al., The Lancet Oncology, 2023)
90 % less surgical diagnostic procedures
Over 50% false positive
on ultrasound

The gray area indicates the proportion (> 55%) of women with peri- or postmenopausal bleeding who need to undergo a surgical diagnostic procedure based on the determination of endometrial thickness by transvaginal sonography, although cancer is present in less than 3% of these women. This is a false positive rate of over 50%.
Less than 3 % false positives
with WID®-easy test

The proportion (5.2%) of women with peri- or postmenopausal bleeding who need to undergo a surgical diagnostic procedure due to a positive WID®-easy test result is shown here in gray. The proportion of false positive results is less than 3%. That’s a 95% reduction in false positives.
Extrapolated to 100 peri- and postmenopausal women presenting with abnormal bleeding, transvaginal sonography (“TVS”) will exceed the 3-mm threshold for endometrial thickness in 55 women. According to the S3 guidelines, all these women must undergo a surgical diagnostic workup (Tao Brush™, Pipelle™, hysteroscopy or curettage). More than 50 of these women will be found not to have endometrial or cervical cancer. Thus, a false positive result was present.
Because of the higher specificity of the WID®-easy test (97.3% WID®-easy / 45.8% TVU), which results in a significantly higher positive predictive value compared with transvaginal ultrasound (PPV 50.0% WID®-easy / 4.9% TVU), the WID®-easy test suggests invasive workup in only five women. That’s a 90% reduction in invasive procedures. Half of these women will be diagnosed with cancer in the histological examination.
The false positive rate is twenty times higher with ultrasound than with the WID®-easy test (52.6% vs. 2.6%). Using the WID®-easy test can therefore avoid 95% of invasive diagnostic procedures in women who are ultimately found not to have endometrial cancer.
Sensitivity, the negative predictive value (NPV) and the rate of false negative results are comparable for both methods.
(Evans et al., The Lancet Oncology, 2023)
Robustness and complications
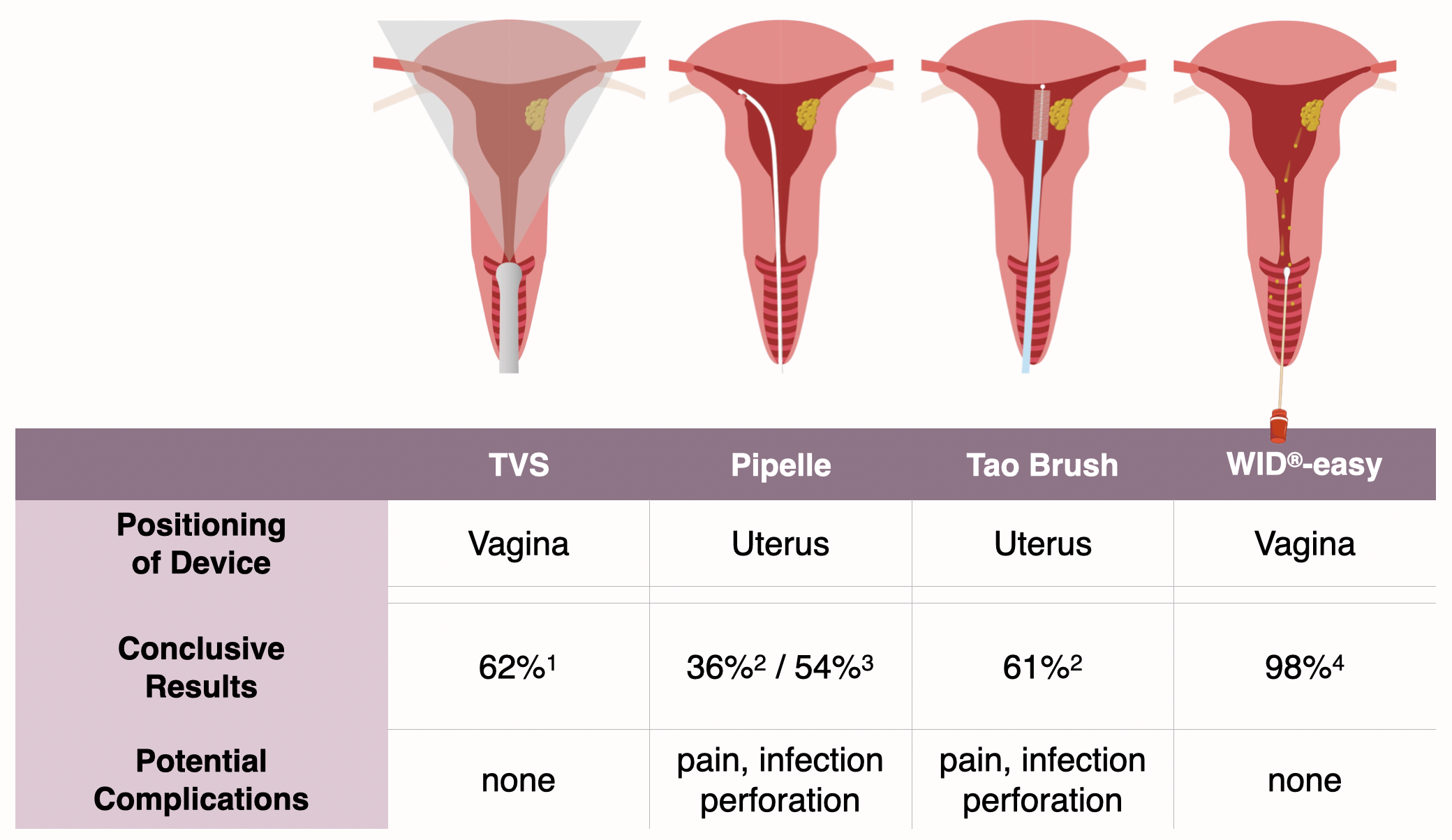
WID®-easy test compared to TVS, Pipelle™ and Tao Brush™
In contrast to examinations using transvaginal sonography, Pipelle™ or Tao Brush™, performing the WID®-easy test almost always (98%) leads to a clear result and no complications are to be expected.
In addition to the convincing performance of the WID®-easy test (sensitivity, specificity, NPV, PPV), the test is also characterized by superior robustness. Furthermore, it can be assumed that the test will not lead to any complications.
In almost 40% of cases, transvaginal sonography and the Tao Brush™ must be expected not to yield a conclusive result. In the Pipelle™ test, the expected value for the proportion of unambiguous results tends to be below 50%.
Performing the WID®-easy test, on the other hand, almost always leads to an unambiguous result (98 %).
Pipelle™ and Tao Brush™ examinations require insertion of the instruments through the cervical canal into the uterus. This can result in pain or infection, and in some cases even perforation of the uterus.
Endocervical carcinoma
Endocervical carcinoma
Endocervical carcinomas cannot be detected during visual inspection using a speculum. Furthermore, these do not lead to an increased endometrial thickness and also remain inconspicuous in cytology.
In a case-control study, the WID®-easy test detected 21 of 22 cervical carcinomas, giving it a sensitivity of 95 %
(Schreiberhuber et al., International Journal of Cancer, 2022).
More conservative treatments
When endometrial cancer is suspected, rapid workup is critical to maximize survival if endometrial cancer is indeed present (Sud et al., The Lancet Oncology, 2020).
In case of a negative WID®-easy test, conservative treatment of women with abnormal bleeding and active monitoring with transvaginal ultrasound and the WID®-easy test is a real option.
You spare your patient a surgical intervention in the hospital and remain in close contact with your patient as her primary contact person during the conservative treatment.

Scientific publications on the WID®-easy test
Evans et al. (2023)
Evans, I., Reisel, D., Jones, A., Bajrami, A., Nijjar, S., Solangon, S. A., Arora, R., Redl, E., Schreiberhuber, L., Ishaq-Parveen, I., Rothärmel, J., Herzog, C., Jurkovic, D. & Widschwendter, M. (2023). Performance of the WID-qEC test versus sonography to detect uterine cancers in women with abnormal uterine bleeding (EPI-SURE): a prospective, consecutive observational cohort study in the UK. The Lancet Oncology, 24(12), 1375-1386. https://doi.org/10.1016/s1470-2045(23)00466-7
Real-life comparison to the state of the art
A prospective study was conducted to compare the performance of the current improved version of the WID-qEC test with the standard procedure in the United Kingdom. The study took place at UCL-Hospital in London.
400 women aged 45 years and older with peri- or postmenopausal abnormal bleeding participated in the study.
The WID-qEC test reduces the number of women who need surgical diagnostic procedures such as hysteroscopy and curettage, even though they do not have cancer, by 87% compared to the standard procedure in the UK and by 95% compared to the standard procedure in Germany, Switzerland and Austria.
The WID-qEC test speeds up diagnosis in women with symptoms of endometrial cancer, leading to better outcomes and healthcare cost savings.
Herzog et al. (2022)
Herzog, C., Marín, F., Jones, A., Evans, I., Reisel, D., Redl, E., Schreiberhuber, L., Paytubi, S., Pelegrina, B., Carmona, Á., Peremiquel-Trillas, P., Frias-Gomez, J., Pineda, M., Brunet, J., Ponce, J., Matias-Guiu, X., De Sanjosé, S., Alemany, L., Olaitan, A., . . Widschwendter, M. (2022). A Simple Cervicovaginal Epigenetic Test for Screening and Rapid Triage of Women With Suspected Endometrial Cancer: Validation in Several Cohort and Case/Control Sets. Journal Of Clinical Oncology, 40(33), 3828-3838. https://doi.org/10.1200/jco.22.00266
Development and validation in five different settings
This publication describes the development and validation of the WID-qEC test.
The test was developed using 726 cervicovaginal smears from women with and without endometrial cancer and validated in 562 unrelated cervicovaginal specimens using three different collection methods and two diagnostic and two predictive settings.
Schreiberhuber et al. (2022)
Schreiberhuber, L., Herzog, C., Vavourakis, C. D., Redl, E., Kastner, C., Jones, A., Evans, I., Zikan, M., Cibula, D., Widschwendter, P., Pfau, K., Math, B., Seewald, M., Amory, S., Obrist, P. & Widschwendter, M. (2022). The WID-qEC test: Performance in a hospital-based cohort and feasibility to detect endometrial and cervical cancers. International Journal Of Cancer, 152(6), 1269-1274. https://doi.org/10.1002/ijc.34275
Real-life cohort and validation for cervical cancer.
WID-qEC test was validated in a real-life hospital cohort of 304 women.
In addition, a case-control study showed the superiority of the WID-qEC test in the detection of cervical carcinoma, especially endocervical carcinoma, compared to cytological examination.
Redl et al. (2024)
Redl, E., Schreiberhuber, L., Ishaq, I., & Widschwendter, M. (2024). High-throughput implementation of the WID-qEC Test: DNA methylation-based test for the detection of uterine cancers. Wiley Analytical Science.
Development of the high-throughput analytics
The article describes the technical implementation and the analytical verification of the WID-qEC test.
The described workflow includes not only the qPCR analysis itself but also the entire test process, including sample collection, automated DNA isolation, quantification and normalization and automated bisulfite conversion as well as downstream analysis and data interpretation.
This process allows laboratories to process a large number of samples exceptionally efficiently.
Illah et al. (2024)
Illah, O., Scott, M., Redl, E., Barrett, J. E., Schreiberhuber, L., Herzog, C., Vavourakis, C. D., Jones, A., Evans, I., Reisel, D., Chandrasekaran, D., Doufekas, K., Graham, R., Kotsopoulos, I. C., MacDonald, N., Arora, R., Olaitan, A., Rosenthal, A. & Widschwendter, M. (2024). High performance of the DNA methylation-based WID-qEC test for detecting uterine cancers independent of sampling modalities. International Journal Of Cancer, 155(5), 800-806. https://doi.org/10.1002/ijc.35000
Comparison of different sampling methods
The study investigated whether different sampling methods influence the validity of the test. Specifically, it compared Cervex brush with PreservCyt against FLOQSwab with eNAT, physician-performed sampling with patient self-sampling, and the exact sampling site (vaginal, cervico-vaginal, or cervical) to assess their influence on the test’s performance.
The results demonstrated that the test results remained consistent irrespective of the sampling method, even for samples stored at room temperature for seven days. While self-sampling led to high specificity, the sensitivity was somewhat lower.
Overall, the WID-qEC test offers a simple, objective and non-invasive method for assessing the risk of malignancy in women with abnormal bleeding.
Ken-Amoah et al. (2024)
Ken-Amoah, S., Redl, E., Domson, B. K. S., Barrett, J. E., Schreiberhuber, L., Herzog, C., Arora, R., Jones, A., Evans, I., Reisel, D., Lamptey-Mills, E., Nachinab, V. B., Pepera, T., Olaitan, A., Obiri-Yeboah, D., Akakpo, P. K. & Widschwendter, M. (2024). Performance of the WID-qEC test to detect uterine cancers in black women with abnormal uterine bleeding: A prospective observational cohort study in Ghana. International Journal Of Cancer, 156(5), 1055-1064. https://doi.org/10.1002/ijc.35260
Performance of the WID-qEC test to detect uterine cancers in black women with abnormal uterine bleeding
The authors investigated the ability of WID-qEC testing to detect endometrial and cervical cancer in black women in Ghana who presented with abnormal uterine bleeding.
The WID-qEC test successfully identified 100% of endometrial and cervical cancers in the study population. The ease of sample collection and low-cost, high-throughput PCR-based analysis makes the WID-qEC test a promising tool for detecting endometrial cancer in all women in low- and middle-income countries and in ethnic groups where sonography performance is known to be particularly poor.
Redl et al. (2026)
Redl, E., Herzog, C., Vavourakis, C., Barrett, J., Jones, A., Evans, I., Reisel, D., Manchanda, R., Bjørge, L., Zikan, M., Cibula, D., Alkasalias, T., Rådestad, A. F., Gemzell-Danielsson, K., Dubeau, L., MacDonald, N., Jurkovic, D., Pashayan, N. & Widschwendter, M. (2026). The cervico-vaginal DNA methylation WID-qEC test: An epigenetic marker associated with ovarian cancer in the absence of endometrial and cervical cancer. International Journal Of Cancer, 1-7. doi:10.1002/ijc.70354
Link between WID®-easy positivity and ovarian cancer
The study investigated the positivity of WID®-easy (WID-qEC) in women without cervical or endometrial cancer. Of 1,269 samples, 6.3% were positive. Ovarian cancer was the strongest independent predictor of positivity (adjusted OR 2.93), and a higher number of ovulatory cycles over a lifetime was also independently associated with positivity.
In a modeled symptomatic population with a baseline ovarian cancer prevalence of 0.38%, WID qEC positivity corresponds to an estimated probability of approximately 1.3% – a significant increase from baseline, even if the absolute risk remains low.
Snowsill et al. (2026)
Snowsill, T. M., Sleigh, S., Barrett, J. & Widschwendter, M. (2026). Cost-Minimization Analysis of the Women’s Cancer Risk Identification-Quantitative Polymerase Chain Reaction Test for Endometrial Cancer Epigenetic Biomarker Compared With Transvaginal Ultrasound for Triaging Abnormal Uterine Bleeding. Value in Health. doi:10.1016/j.jval.2026.03.2235
WID®-easy vs. transvaginal ultrasound: A cost minimization analysis in the UK NHS
A cost minimization analysis in the UK National Health Service (NHS) compared the WID®-easy test (WID-qEC) with transvaginal ultrasound (TVUS) as a first-line triage test in women aged 45 and over with abnormal uterine bleeding. Using a decision tree model based on the EPI-SURE cohort (n=399) and the NHS Reference Costs 2023/24, only 12% of women required definitive invasive assessment under the WID®-easy strategy compared to 57% under the TVUS strategy.
The break-even price for cost equivalence was £302 per test. At an indicative price of £200, the probability of cost savings exceeded 99%; at £150 there were mean savings of £152 per patient. Across BMI and ethnic subgroups, break-even prices remained stable (£290-£331). The chosen cost minimization approach does not attribute value to the reduction in pain, anxiety and diagnostic delay – effects that a reduced number of hysteroscopies and endometrial biopsies would bring to the overburdened NHS system, where urgent referrals for suspected cancer have more than tripled in recent years (2009-2023).
Olaitan et al. (2026)
Olaitan, A., Pashayan, N., Butler, J., Cibula, D., Costas, L., Crosbie, E. J., Falconer, H., Fotopoulou, C., Akapo, P. K., MacDonald, N., Moss, E., Mueller, M., Rosenthal, A. N., Sundström, K., Hillemanns, P., Manchanda, R., Sleigh, S. & Widschwendter, M. (2026). Transforming Endometrial Cancer Diagnosis: The Case for Molecular Triage in Abnormal Uterine and Postmenopausal Bleeding. International Journal of Gynecological Cancer. doi:10.1016/j.ijgc.2026.104772
Endometrial cancer diagnostics on the brink of change: a plea for molecular triage
An international review in the International Journal of Gynecological Cancer criticizes the current diagnostic pathway for abnormal uterine bleeding. With a cancer prevalence of only 3%, the standard pathway (transvaginal ultrasound + hysteroscopy/biopsy) leads to around 33 biopsies per carcinoma detected, misses ~25% of the worst prognostic serous tumors and is significantly weaker in black women.
Molecular triage is being discussed as an alternative. The WID-qEC test (WID®-easy) achieved a sensitivity of 90.9 %, specificity of 92.1 % and an NPV of 99.7 % in the EPI-SURE cohort (n=399). With 3,500 annual referrals, urgent hysteroscopies were reduced from 1,850 to 210 – with detection of over 95% of all carcinomas.
Are you interested in the WID®-easy test?
In the European Union and Switzerland, the WID®-easy test is manufactured and used by healthcare facilities, such as diagnostic laboratories, in accordance with the European In Vitro Diagnostics Regulation (IVDR) Article 5, paragraph 5, or corresponding national regulations.
In the UK, the WID®-easy test is labelled in accordance with the UK Medical Devices Regulations 2002 (SI 2002 No. 618, as amended) (UK MDR 2002)
Partners who currently offer the WID®-easy test can be found on the map below. Please contact one of these providers directly to obtain the necessary documents and materials for commissioning, sampling and logistics.

Advanced Global Health Ltd
30 Great Guildford Street, London, Great Britain
Tel: +44 207 078 8867
Mail: info@aghealth.co.uk
Web: www.aghealth.co.uk

European Oncology Lab GmbH
Klaus-Tussing-Str. 2 / Gesundheitspark 2, 66386 St. Ingbert, Germany
Tel: +49 6894 95310
Mail: labor@europeanoncologylab.com
Web: www.europeanoncologylab.com

labor team w ag
Blumeneggstrasse 55, 9403 Goldach, Switzerland
Tel: +41 71 844 45 45
Mail: info@team-w.ch
Web: www.laborteam.ch

Laboratory - Doz. DDr. Stefan Mustafa
Ziehrerplatz 9, 1030 Vienna, Austria
Tel: +43 1 713 91 88
Mail: office@labor-mustafa.at
Web: www.labor-mustafa.at

Medilab GmbH
Strubergasse 20, 5020 Salzburg, Austria
Tel: +43 662 2205
Mail: office@medilab.at
Web: www.medilab.at

NextGen Molecular Supplies
Incubation Drive 8, 2021 Midrand, South Africa
Tel: +27 87 148 9309
Mail: customer_care@ngms.co.za
Web: www.ngms.co.za

UAB Genotipas
Aviečių g. 14, 08418, Vilnius, Lithuania
Tel: +370 686 55909
Mail: info@genotipas.lt
Web: www.genotipas.lt






